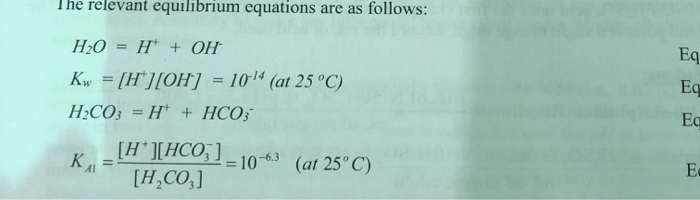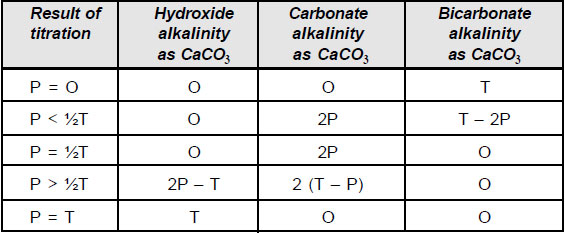
Determination of Alkalinity of Water | Environmental Science and Engineering Laboratory Methodology | Biocyclopedia.com

SOLVED: Calculate the exact alkalinity of water having 1.60×10^-8 mol/L of carbonate ion, 1.20×10^-4 mol/L of bicarbonate ion, and a pH of 5.5. State your answer in units of mg/L as CaCO3.
![SOLVED: 6. Calculate the total hardness, calcium hardness and magnesium hardness for a water sample having a pH of 7.2 and the following concentrations of ions: [Ca ] = 40 mgL [Mg ] = SOLVED: 6. Calculate the total hardness, calcium hardness and magnesium hardness for a water sample having a pH of 7.2 and the following concentrations of ions: [Ca ] = 40 mgL [Mg ] =](https://cdn.numerade.com/ask_previews/32bca3e4-ee47-44d7-8fb8-7e86804add2d_large.jpg)
SOLVED: 6. Calculate the total hardness, calcium hardness and magnesium hardness for a water sample having a pH of 7.2 and the following concentrations of ions: [Ca ] = 40 mgL [Mg ] =